DOSING
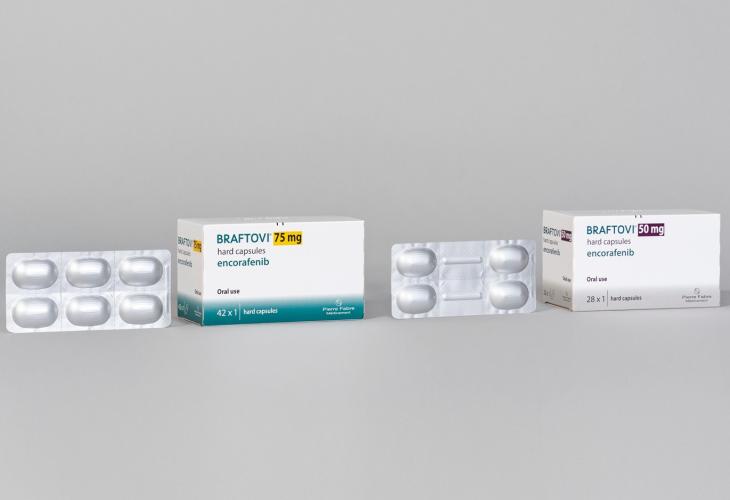
BRAFTOVI® presentation
BRAFTOVI® presentation
BRAFTOVI® is available in packs of:
| BRAFTOVI® 75 mg | 42 x 1 hard capsules and 168 x 1 hard capsules* |
| BRAFTOVI® 50 mg | 28 x 1 hard capsules |
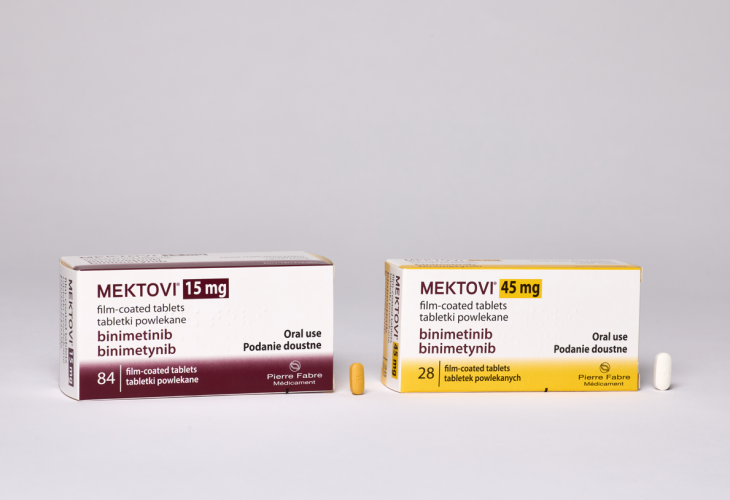
MEKTOVI® presentation
MEKTOVI® presentation
MEKTOVI® is available in packs of:
| MEKTOVI® 15 mg | 84 film-coated tablets |
| MEKTOVI® 45 mg* | 28 film-coated tablets |
Recommended dosing
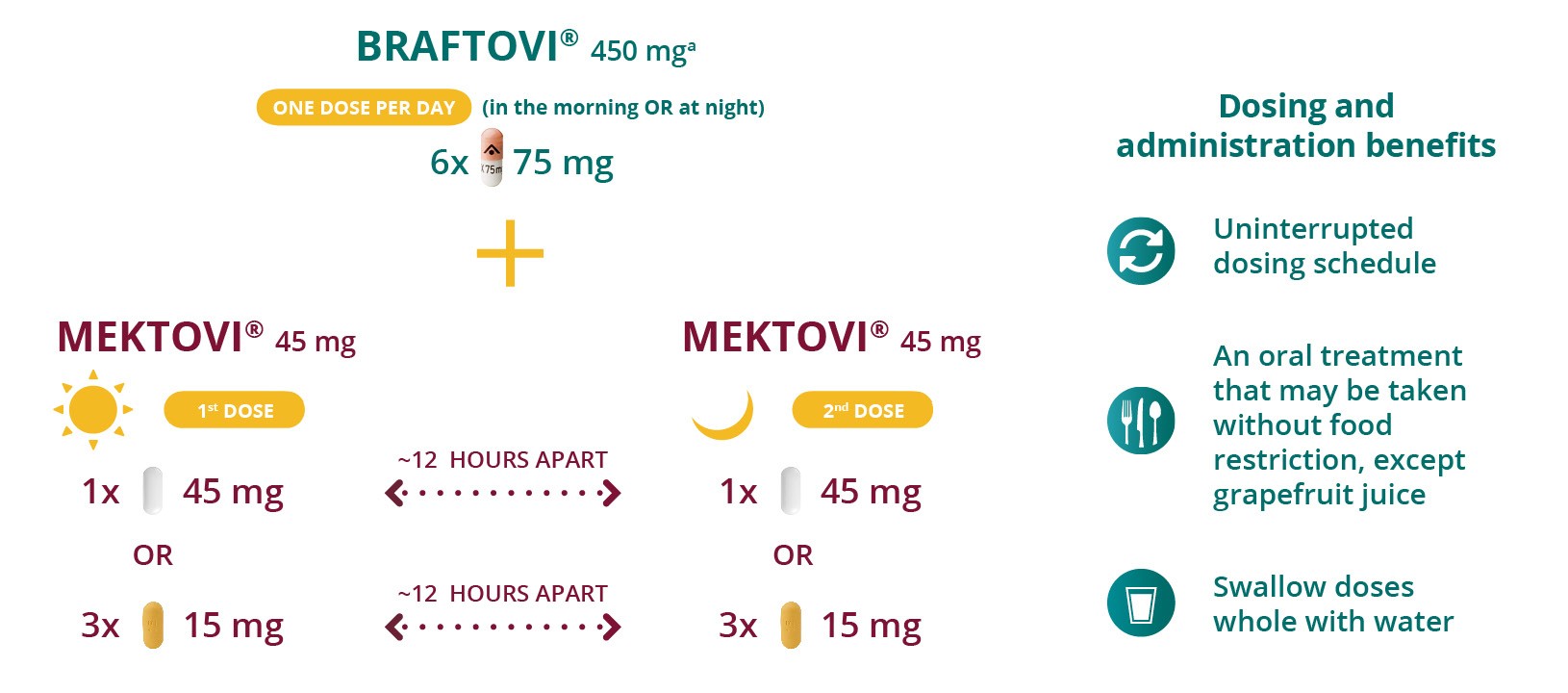
Treatment with BRAFTOVI® + MEKTOVI® should be continued until the patient no longer derives benefit or the development of unacceptable toxicity.
a For patients with mild hepatic impairment, administration of BRAFTOVI should be undertaken with caution at a reduced dose. In the absence of clinical data, BRAFTOVI is not recommended in patients with moderate to severe hepatic impairment.1
Adapted administration methods for patients unable to swallow1,2
BRAFTOVI 75 mg and 50 mg capsulesa,b
May be opened and the content dispersed in a small quantity (~20 mL) of apple sauce and taken immediately1

MEKTOVI 15 mg tablets onlyc
May be dispersed in a small glass (~10 mL) of either water, orange juice or apple juice and taken immediately2
The glass should be rinsed with ~10 mL of water, orange juice or apple juice, and content drunk immediately2

Patient monitoring is recommended prior, during and after the treatment. For more information on safety, explore our Therapy Management Guide, on the Support and Resources page.
Recommended dose adjustments
BRAFTOVI® + MEKTOVI® are indicated to be taken in combination.
The management of ARs may require dose reduction, temporary interruption, or treatment discontinuation.

Dose modifications are recommended to manage certain adverse reactions.
Please see Section 4.2 of the full Summaries of Product Characteristics for additional information.
aThere are limited data for dose reduction to 100 mg QD. If unable to tolerate 100 mg QD, permanently discontinue BRAFTOVI.1
bIf unable to tolerate 30 mg BID, permanently discontinue MEKTOVI.1
BID, twice daily; QD, once daily.
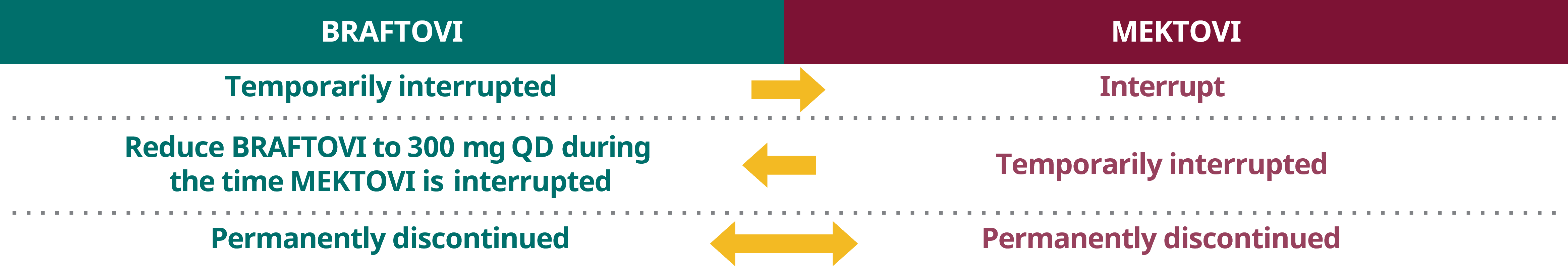
Dose interruption and discontinuation
When temporarily interrupting or permanently discontinuating BRAFTOVI or MEKTOVI, please adapt the other drug's posology according to the table below.
For complete information, please refer to the Summaries of Product Characteristics.